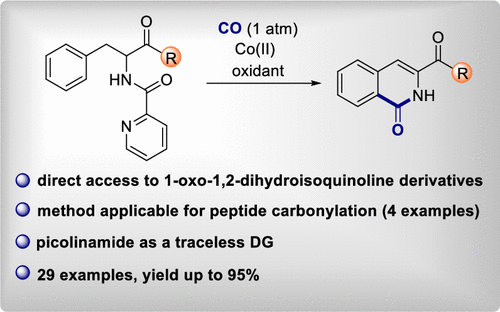
Continuing studies of cobalt-catalyzed C(sp2)–H carbonylation reactions Liene Grigorjeva et al. (Organic Synthesis Methodology group) have reported an efficient protocol for the synthesis of 1,2-dihydroisoquinolinone derivatives based on an inexpensive cobalt(II) salt catalyst. This method is published in the high-impact ACS journal Organic Letters (IF=6.555).
In this approach carbonylation of amino acids was accomplished using picolinamide as a traceless directing group, CO (1 atm) as the carbonyl source, and Co(dpm)2 as the catalyst. The authors demonstrated that a broad range of phenylalanine derivatives bearing diverse functional groups were tolerated. The method can be successfully applied for the C(sp2)–H carbonylation of short peptides as well.
Read the article:
Lukasevics, L., Cizikovs, A., Grigorjeva, L.*
Cobalt-Catalyzed C(sp2)–H Carbonylation of Amino Acids Using Picolinamide as a Traceless Directing Group.
Org. Lett., 2021. DOI: 10.1021/acs.orglett.1c00660
This research is funded by the Latvian Council of Science, project [Cobalt catalyzed C–H bond functionalization], project no. lzp-2019/1-0220.