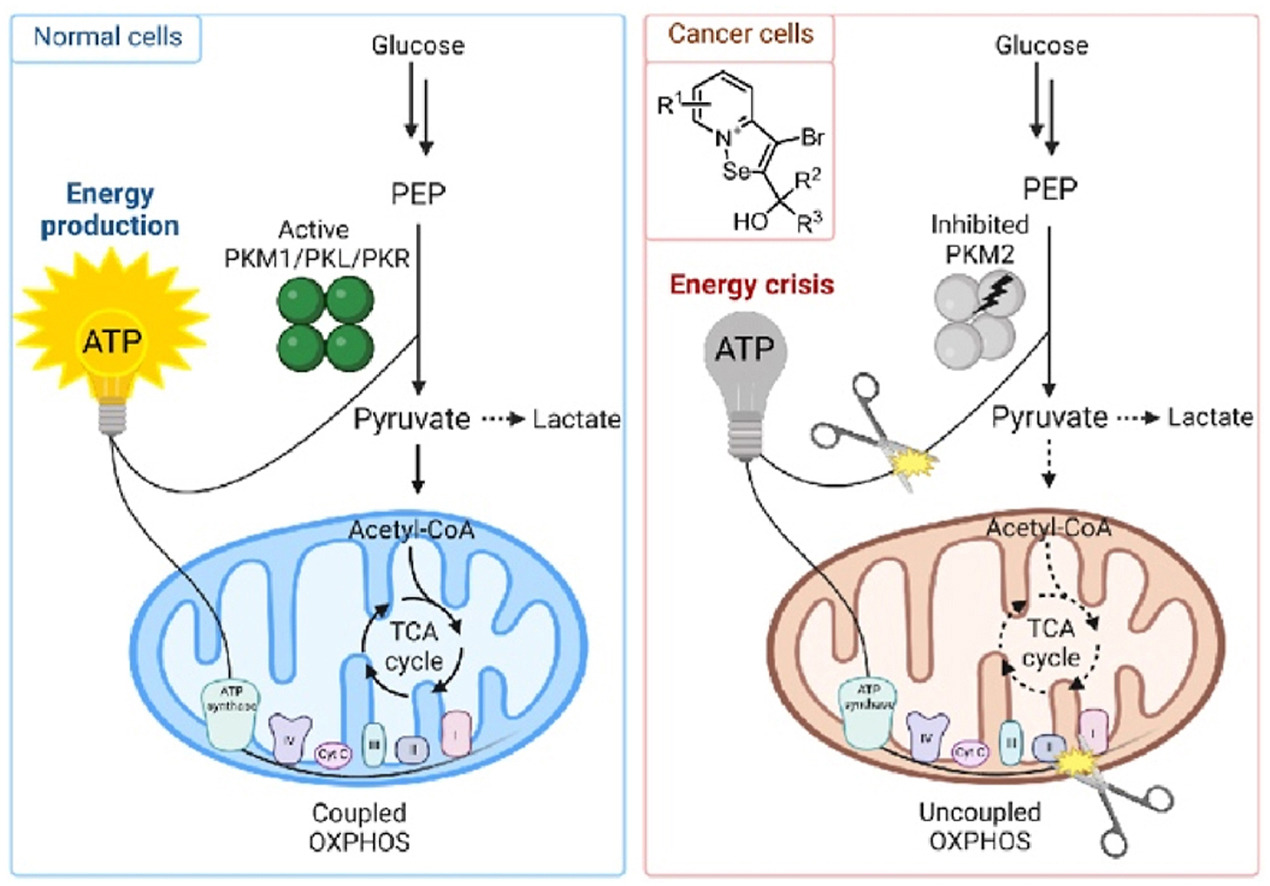
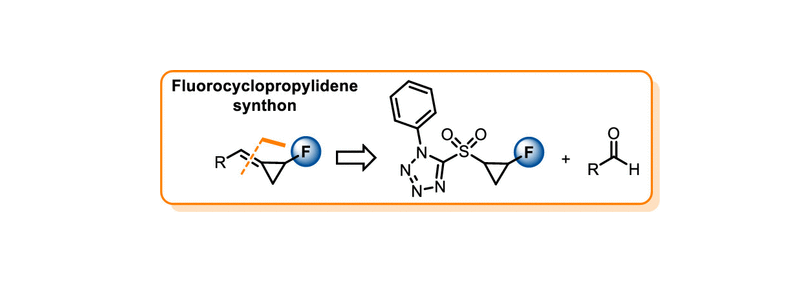
EU-funded Project to Pioneer a State-of-the-Art Enzyme Technology Ecosystem Across Europe to Foster Competitive Research and Innovation
Enzyme technology stands as a beacon of innovation with the potential to revolutionise industries across the globe. Recognising the transformative power of this technology, a