Prof. Edgars Suna’s group (Latvian Institute of Organic Synthesis) has elaborated a new synthetic approach to obtain densely substituted enantiopure cyclic sulfinamides. The method has been published in the journal Organic Letters (IF=6.005).
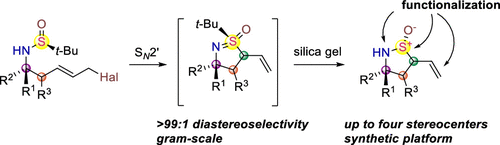
This synthetic approach toward densely substituted enantiopure cyclic sulfinamides possessing up to four consecutive stereogenic centers was developed based on a completely diastereoselective SN2′ cyclization/tert-Bu cleavage sequence. Researchers demonstrated diastereospecific transformation of the obtained scaffold into chiral SVI derivatives such as sulfoximines and sulfonimidamides as well.
Read the article:
Jersovs, G.; Bojars,M.; Donets, P.A.; Suna, E.*
Synthetic Approach toward Enantiopure Cyclic Sulfinamides.
Org. Lett., 2022. DOI: 10.1021/acs.orglett.2c01738