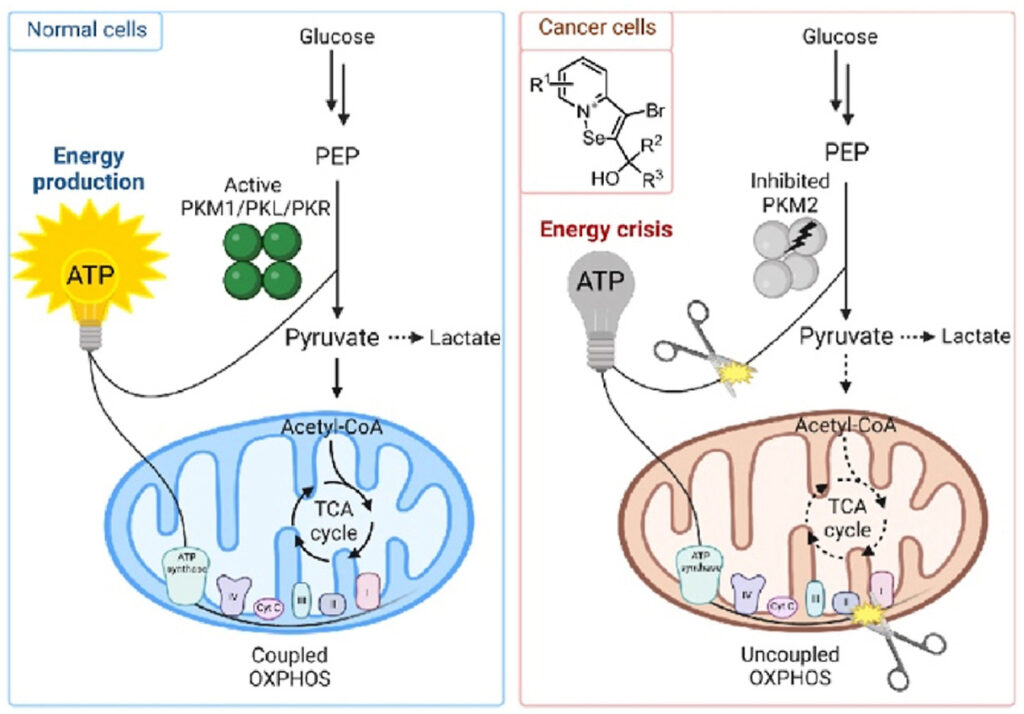
A novel class of selective PKM2 inhibitors as anti-cancer agents and their molecular mechanism of action have been discovered by Pavel Arsenyan (LIOS, Pharmacomodulators Synthesis group) and his group. The study has been published in the European Journal of Medicinal Chemistry (IF = 7.088).
Researchers have discovered a novel series of potent and isoform-selective PKM2 inhibitors bearing a carbonyl- [1,2]isoselenazolo[2,3-a]pyridinium moiety. They found that, unlike other PKM2 inhibitors, isoselenazolium derivatives reduce the proportion of dimeric PKM2 species and induce an unstable PKM2 conformation that results in impaired enzymatic function. Moreover, PKM2 is inhibited both at the protein and transcript level, as was shown by the reduction in PKM2 mRNA. This inhibitory activity leads to intracellular redox imbalance and, consequently, to cell death. These findings is providing a basis for the novel class of drug candidates with a unique molecular mechanism and reveal specific structural features of PKM2. Furthermore, a potent and stable inhibitor would be an integral part of future studies focusing on the metabolic role of PKM2 in oncogenesis.
Read the article
Dimitrijevs, P., Makrecka-Kuka, M., Bogucka, A., Hyvönen, M., Pantelejevs, T., Arsenyan, P.*
Development of isoselenazolium chlorides as selective pyruvate kinase isoform M2 inhibitors.
Eur. J. Med. Chem. 2023, 257, 115504. DOI: 10.1016/j.ejmech.2023.115504