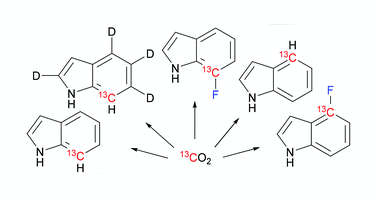
Ansis Maleckis (Latvian Institute of Organic Synthesis), Iresha D. Herath and Gottfried Otting (Australian National University) have demonstrated the use of 13C labeled indoles and fluoroindoles for in vitro protein synthesis.
In the cell-free reaction medium indole is converted to the corresponding tryptophan which is then incorporated into the structure of the target protein. Incorporation of 13C–1H and 13C–19F spin pair labeled indoles into proteins enables high-resolution and high-sensitivity nuclear magnetic resonance (NMR) spectroscopy. The synthesis of indoles labeled with 13C–1H and 13C–19F spin pairs utilize inexpensive carbon-13C dioxide as the 13C isotope source and ruthenium-mediated ring closing metathesis is employed for the construction of the indole carbocycle.
This research is published in the Organic & Biomolecular Chemistry (IF = 3.412).
Read the article:
Ansis Maleckis*, Iresha D. Herath, Gottfried Otting*
Synthesis of 13C/19F/2H labeled indoles for use as tryptophan precursors for protein NMR spectroscopy.
Org. Biomol. Chem., 2021, DOI: 10.1039/D1OB00611H