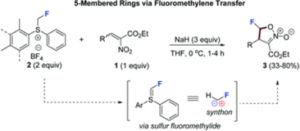
Janis Veliks et al. (Organic Synthesis Methodology group) have reported a new strategy of synthesizing monofluorinated five-membered rings via fluoromethylene transfer. This approach has been published in the journal Organic & Biomolecular Chemistry (IF=3.412).
This work demonstrates for the first time that one-fluorine-one-carbon modification of a substrate could be a viable strategy of accessing monofluorinated five-membered rings. The synthetic methodology was developed to access monofluorinated isoxazoline-N-oxides in one step starting from substituted 2-nitroacrylates using fluoromethylsulfonium reagents.
Read the article:
Sperga, A.; Kaziaa, A.; Veliks, J.
Monofluorinated 5-membered rings via fluoromethylene transfer: synthesis of monofluorinated isoxazoline N-oxides.
Org. Biomol. Chem., 2021. DOI: 10.1039/D1OB00270H.