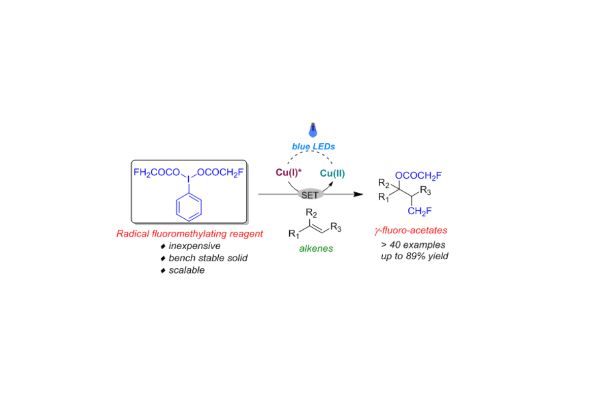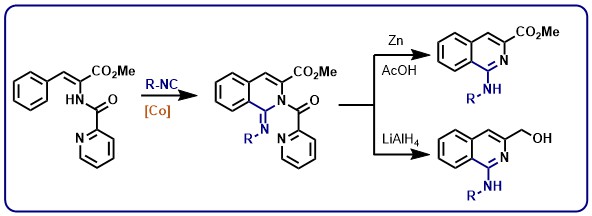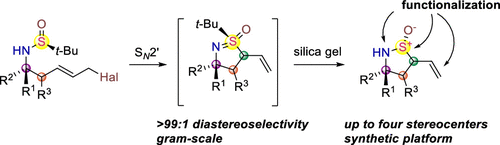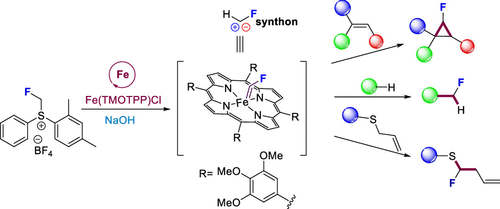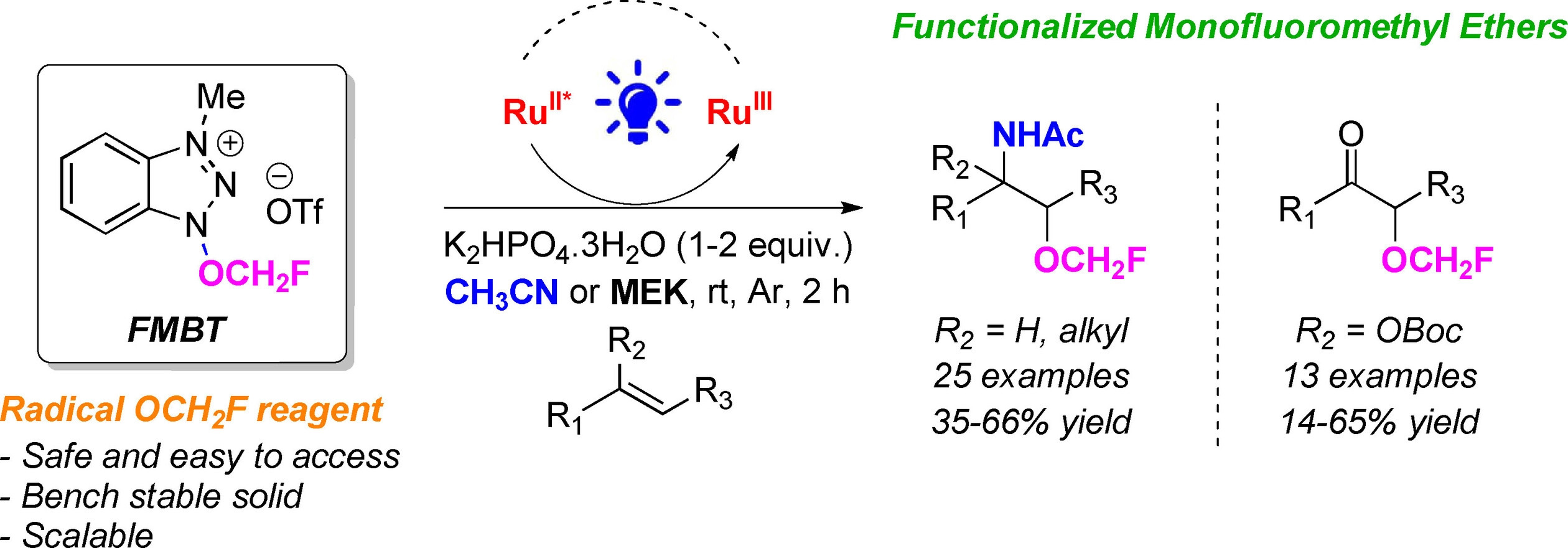
A convenient synthesis method for obtaining functionalized monofluoromethyl ethers has been elaborated by Janis Veliks’ group
Fluoroalkyl ethers play a significant role in various fields such as pharmaceuticals, agrochemicals, nuclear imaging, and material sciences. Due to their importance, it is crucial