Fluoroalkyl ethers play a significant role in various fields such as pharmaceuticals, agrochemicals, nuclear imaging, and material sciences. Due to their importance, it is crucial to develop new synthetic methods for the synthesis of these compounds.
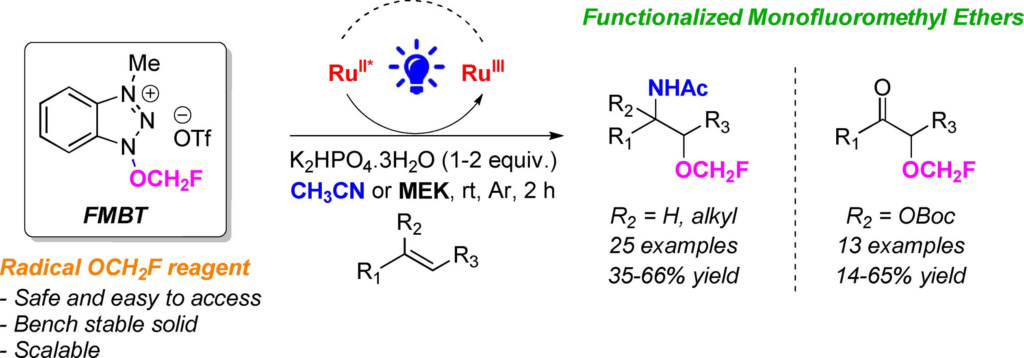
Researchers at the Organic Synthesis Laboratory (LIOS), led by Janis Veliks, have developed a convenient synthesis method to obtain functionalized monofluoromethyl ethers using easy-to-access redox-active N–O reagent, 1-(fluoromethoxy)-3-methyl-benzotriazolium triflate (FMBT), serving as a radical source of (mono)fluoromethoxy group under photocatalytic conditions.
They have demonstrated the synthetic potential of FMBT in the functionalization of olefins to access amino and keto-monofluoromethyl ethers, benefiting the related study in the context of chemical biology and drug discovery.
The elaborated methodology has been published in the journal Advanced Synthesis & Catalysis (IF= 5.98).
Read the article:
Ramkumar, N., Sperga, A., Belyakov, S., Mishnev A., Zacs, Dz., Veliks, J.*
Visible-Light Photoredox-Catalyzed Radical Fluoromethoxylation of Olefins
Adv. Synth. Catal. 2023. DOI: 10.1002/adsc.202300130